Electrolysis Of Water
Water is vital for every living organism that we know about, and if humans are ever to set up permanent residence away from the Earth we'll need to make sure that we have a good supply of it. Fortunately, we now know that water is fairly plentiful in our Solar system (although most of it is in the form of ice). In fact some of Saturn's and Jupiter's moons could be more ice than rock.
It isn't just useful for drinking and growing food though. Water is a chemical compound of hydrogen and oxygen, and you can split it into rocket fuel or air to breathe using electrolysis - basically just passing electricity through it:

The basic experiment
You can easily do this at home using readily available equipment:
- A container to hold the water
- A 9V battery
- A pencil - cut in half, and with each piece sharpened at both ends.
- A plastic ruler
- Some elastic bands
- A ball-point pen - the type with a transparent plastic case. Make sure it doesn't have a hole in the side.
The main part can be assembled as shown in the picture below; it's based on quite an ingeniously simple set-up from this site.
Fill the container with warm water, and place the ruler in at an angle so that the ends of the pencils are submerged, but the battery is not. You should see bubbles start to appear almost immediately. Congratulations, you're doing science!
You should see that one of the pencils is producing twice as many bubbles as the other. If you revisit the equation at the top of the page, then it should become clear which gas is which.
You can increase the rate of electrolysis by adding an electrolyte. This will help the electricity flow more easily between the pencil tips. The most easily available electrolyte in the home is salt - just add some to the water. Beware, depending on how much salt you add the anode will now produce some chlorine gas, following the equation below:

NaOH is better known as caustic soda (sodium hydroxide), and is soluble in water so you will not see it. Chlorine is a gas that can cause irritation - used as a weapon in World War I. The amount of chlorine produced here will be small enough to not to be harmful, but you should still work in a well ventilated area.
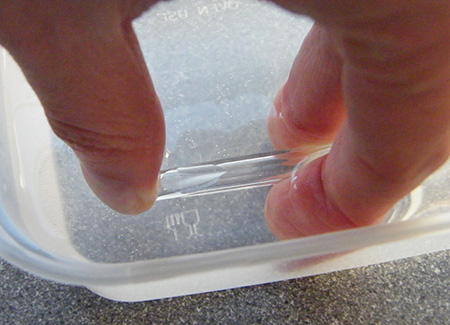
You can use the casing of a ball-point pen to collect the gases. Submerge it in the water at an angle so that the air escapes, and then use your thumb to make one of the ends air-tight. When you tip up the casing, so long as the open end remains under the water, it will remain full. Be extra careful if collecting the mixture of oxygen and chlorine.
Place the open end of the pen casing over the end of the pencil to collect the gas. Be sure to keep your thumb tightly over the opposite end of the pen casing.
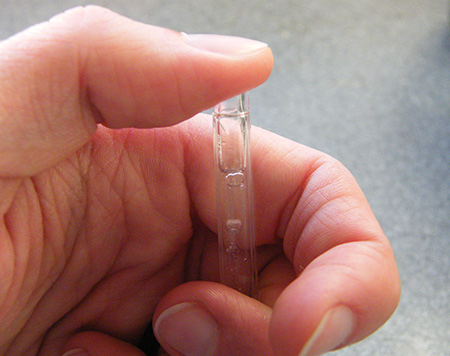
You will soon start to see a pocket of gas appearing at the top of the casing. It could take 15 minutes or more to fill the tube, but after a couple of minutes you will have enough gas to test.
Before taking the casing out of the water, use your other hand to block the bottom end.
A party candle wedged into a a firm platform (such as an over-ripe banana) serves as a good flame. Any stable source of fire will be suitable. Simply move the gas end towards the flame, and remove your finger. Hydrogen will "explode", which translates into a faint pop. This particular set-up does not work well for identifying oxygen (because it is roughly the same weight as the surrounding air, and less likely to escape from the top), but it would make the flame burn more intensely.
How does NASA do it?
Although this system works, it isn't the best configuration. One of the excellent activities [that used to be] at NASA's Space Station Live! site was the ability to wander around mission control, and examine the monitoring terminals. If you made your way to the ETHOS terminal, and clicked on the water molecule on top, then you could find out about how the professionals do it.
One of the main differences is the choice of electrolyte, for which NASA uses a chemical called potassium hydroxide (KOH). Its main benefit is that it increases the rate of reaction, but you still only get oxygen and hydrogen released.
More details
So far the explanation of what's happening, and the equations, have been for the reaction as a whole. In fact it is more accurate to view it as two separate reactions - one taking place at the tip of each pencil. The pencil connected to the negative terminal of the battery (-) is referred to as the cathode, and the pencil connected to the positive terminal of the battery (+) is called the anode.
At the cathode (negative), electrons (e-, which have a negative charge) get added. This is called reduction:

At the anode (positive), electrons are taken out - splitting the water into hydrons and oxygen. This is called oxidation.

The chemicals hydroxide (OH-) and hydron (H+) may be less familiar, but the important thing to notice is their charge. There is a build up of negative ions (charged particles) at the cathode, and positive ions at the anode. Some will linger around the end of the pencils, but some will drift between the pencil ends and cancel out the charge. Further information can be found on the wikipedia page, and at this virtual chemistry lab.
Testing the pH
pH is a measure of how acidic or basic a chemical is. You can buy various electronic testers and colour changing paper strips, but a very good indicator can be made from red cabbage. Simply chop up some red cabbage, and put it in a container with some boiling water. After around 15 minutes drain out the cabbage pieces and you'll be left with a dark purple liquid.
In the image below the plain cabbage juice is in the middle glass. The glass in the background has had a few drops of vinegar added (an acid), while the glass in the foreground has had a small amount of bicarbonate of soda added (a base).
If you use a diluted red cabbage liquid (with some added salt) for your electrolysis, then you will see red appear around one of the pencil ends. If you look closely you should also see the liquid around the other pencil turning more greenish-blue. The image below does not capture the process very well, but it is noticeable to the naked eye.
Why this happens is explained in the "More details" section above. Where there are mainly hydroxide ions, the liquid becomes a base, and where there are mainly hydron ions the liquid becomes acidic. This is another clue as to what is happening.